KASA to Support Generic Drug Review – A Contract Pharma Featured Article
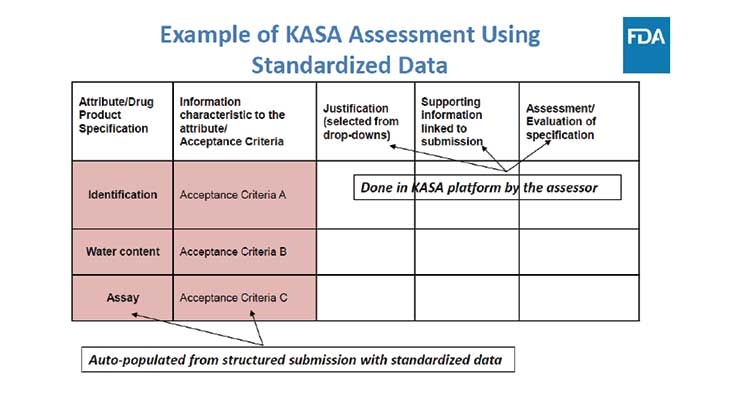
Contract Pharma published an article in April addressing the need for a modernized version of “our generic drug review process.” FDA anticipates a data-based assessment will “improve consistency, transparency, communication, and objectivity of regulatory actions as well as knowledge management within the Agency.” Knowledge-aided Assessment & Structured Application (KASA) is in support of the Generic […]











